Building Agent Studio: How Medable Is Using Agentic AI to Accelerate Clinical Trials

Listen to this episode on: Spotify | Apple Podcasts
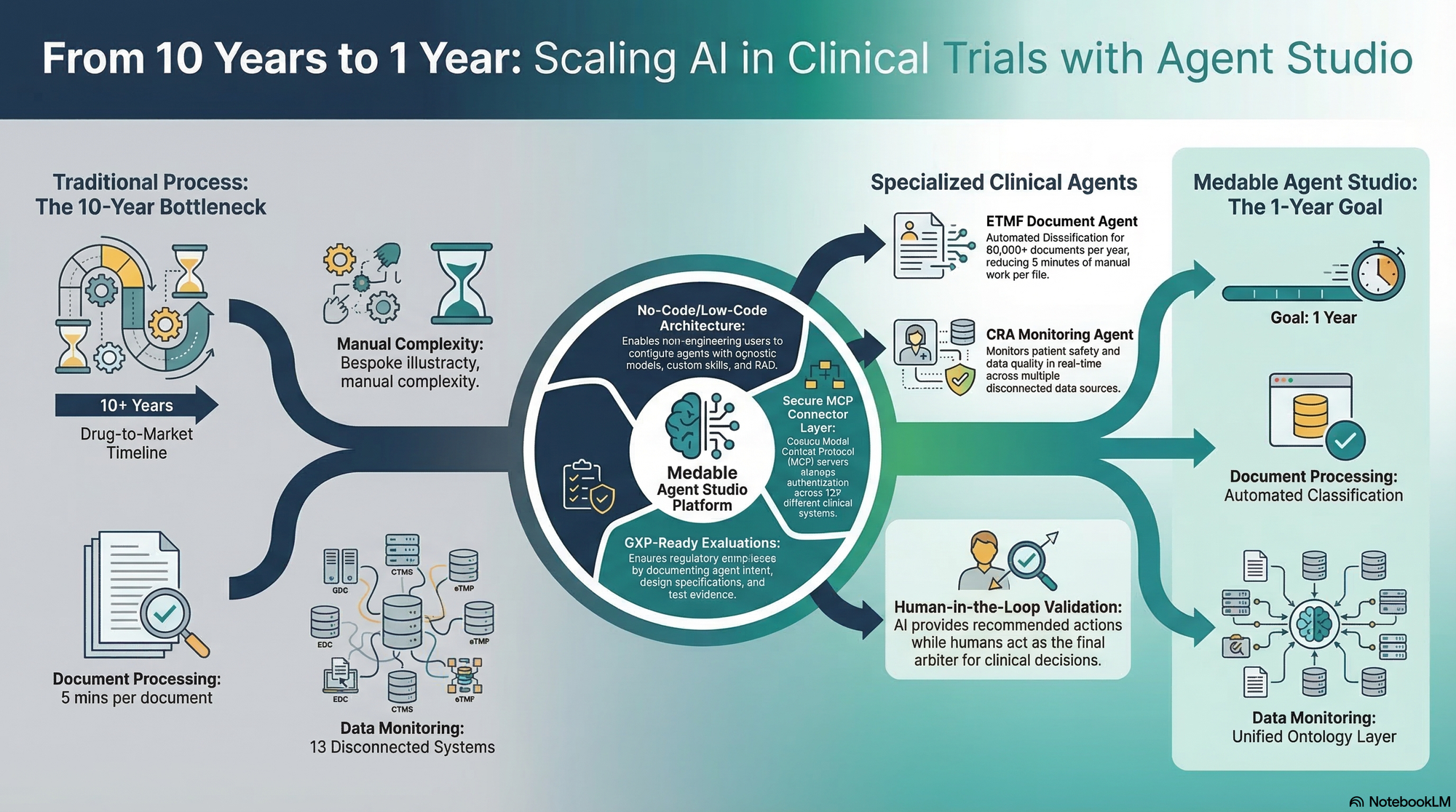
What if AI could help reduce the 10-plus years it takes to get a new drug to market? That's the driving ambition behind Medable's agentic platform—and the bet that led them to build Agent Studio.
In this episode of Just Now Possible, Teresa Torres talks with four members of the Medable team: Luke Bates (Product Leader, Agent Studio), Jen Brown (Product Manager), Matt Schoolfield (Product Designer), and Fiachra Matthews (Principal Architect). Together they share how Medable—a clinical trial platform used by global pharmaceutical companies—built Agent Studio, a no-code/low-code platform for configuring and deploying agents across the clinical trial lifecycle.
You'll hear about the two agents they've built on top of it: an ETMF agent that automates document classification across 80,000-plus documents per year, and a CRA agent that monitors patient safety and data quality across 13 different clinical systems. The conversation goes deep on the architecture behind it all—how they handle RAG and context management at scale, why they built custom MCPs with an authentication layer, how they designed evals for a regulated GXP environment, and what human-in-the-loop really looks like when clinical decisions are on the line.
It's a rare look inside an enterprise AI platform built for one of the most regulated industries in the world—and a team that's still figuring it out in real time.
Show Notes
Guests
- Luke Bates, Product Leader (Agent Studio), Medable
- Jen Brown, Product Manager, Medable
- Matt Schoolfield, Product Designer, Medable
- Fiachra Matthews, Principal Architect, Medable
What we cover in this episode:
- What Medable does: enabling global clinical trials across 100+ languages and accelerating drug-to-market timelines
- The two agents built on Agent Studio—ETMF (document classification) and CRA (clinical data monitoring)—and the problems they solve
- Why Medable chose a platform approach to agents instead of one-off builds
- How Agent Studio works: models, skills, knowledge bases, MCP connectors, versioning, and trigger types
- Three deployment models: Medable-built products, services-led custom builds, and self-serve platform access
- RAG approaches at scale: embeddings vs. markdown hierarchies vs. just-in-time MCP retrieval
- How they built a unified ontology layer to map terminology across 13 different clinical data systems
- Why they built custom MCPs with an authentication and credentialing wrapper
- Context window management with sub-agents and automatic tool filtering
- Evaluation design in a GXP-regulated environment: golden datasets, production monitoring, and the challenge of human feedback as ground truth
- How they document agent intent → specification → test evidence to satisfy regulatory bodies
- The "full self-driving" vision for clinical trials and what it would take to get there
Resources & Links
- Medable - Clinical trial platform powering Agent Studio
Chapters
00:00 Meet The Medable Team
01:14 Medable Mission And Scope
03:27 Agent Studio Platform Overview
06:29 ETMF Document Automation
08:47 CRA Agent For Monitoring
10:40 Clinical Trial Workflow Primer
14:34 Why Build A Platform
17:51 Learning AI As A Team
21:47 Early Days Of Agent Studio
23:15 How Agents Are Built
25:15 Customer Adoption And UX
30:00 Skills And MCP Standards
31:15 Scaling Context Retrieval
33:07 RAG Patterns And Tradeoffs
34:48 Ontology Data Layer Explained
38:01 Customer Friendly Agent Setup
42:19 MCP Security And Connectors
44:36 Tool Bloat And Subagents
50:44 Evals For Reliable Agents
54:40 Human Feedback Isn’t Truth
57:43 GXP Compliance For Agents
01:03:34 Full Self Driving Trials
Full Transcript
Podcast transcripts are only available to paid subscribers.



